|
In this case, the original carboxylic acid, C2H5COOH, is called propanoic a. To give the IUPAC name of it, we replace the “-ic acid” part of its original carboxylic acid with “yl” and then add “chloride”. WebAnswer (1 of 2): This is an acid chloride, which is a derivative compound of carboxylic acid. There is a systematic nomenclature of heterocyclic compounds, but it will not be discussed here.What is the IUPAC name of C2H5CoCl? - Quora Since these names are not based on a rational system, it is necessary to memorize them. Many aromatic and heterocyclic amines are known by unique common names, the origins of which are often unknown to the chemists that use them frequently. These are the names given in the last row (colored black). Finally, a common system for simple amines names each alkyl substituent on nitrogen in alphabetical order, followed by the suffix -amine.These CA names are colored magenta in the diagram. The additional nitrogen substituents in 2º and 3º-amines are designated by the prefix N- before the group name. For 1º-amines such as butanamine (first example) this is analogous to IUPAC alcohol nomenclature (-ol suffix). The Chemical Abstract Service has adopted a nomenclature system in which the suffix -amine is attached to the root alkyl name.dimethylamino in the fourth example) includes the names of all but the root alkyl group. For 2º and 3º-amines a compound prefix (e.g. The simple -NH substituent found in 1º-amines is called an amino group. This system names amine functions as substituents on the largest alkyl group. The IUPAC names are listed first and colored blue.For example, (CH 3) 4N (+) Br (–) is tetramethylammonium bromide. A nitrogen bonded to four alkyl groups will necessarily be positively charged, and is called a 4º-ammonium cation. The third and fourth compounds in the row are 2º and 3º-amines respectively. The first two are classified as 1º-amines, since only one alkyl group is bonded to the nitrogen however, the alkyl group is primary in the first example and tertiary in the second. The four compounds shown in the top row of the following diagram are all C 4H 11N isomers. When applied to amines these terms refer to the number of alkyl (or aryl) substituents bonded to the nitrogen atom, whereas in other cases they refer to the nature of an alkyl group. Furthermore, the terms primary (1º), secondary (2º) & tertiary (3º) are used to classify amines in a completely different manner than they were used for alcohols or alkyl halides. The nomenclature of amines is complicated by the fact that several different nomenclature systems exist, and there is no clear preference for one over the others. If you are uncertain about the IUPAC rules for nomenclature you should review them now.Īmines are derivatives of ammonia in which one or more of the hydrogens has been replaced by an alkyl or aryl group. 
Halogens, on the other hand, do not have a suffix and are named as substituents, for example: (CH 3) 2C=CHCHClCH 3 is 4-chloro-2-methyl-2-pentene. 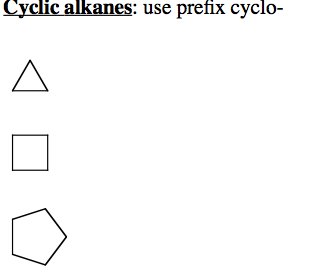 
This is common for the carbon-carbon double and triple bonds which have the respective suffixes ene and yne. 
The presence of the function may be indicated by a characteristic suffix and a location number. In the IUPAC system of nomenclature, functional groups are normally designated in one of two ways.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
